- Products
- Medical Devices
- Materials
- Photochemical Etching
- Secondary Services
- Applications
- Quality Engineering

Aneurysm Prevention Devices
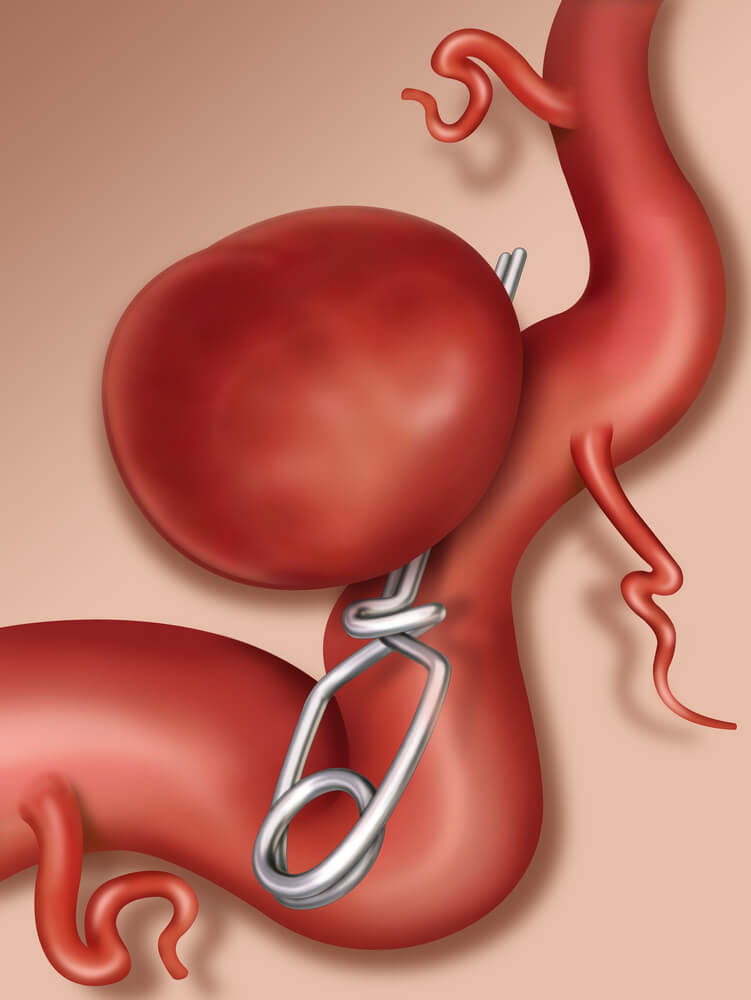
Tecomet Inc. offers precision, high quality aneurysm device etching. Our experienced staff will work with you on your project from research, development and design through FDA approval and commercial launch. The precision of our chemical machining process allows us to produce the intricate features required for these etched aneurysm prevention devices. Tecomet Inc. specializes in etching nitinol, stainless steel and titanium, which are the most common metals used for these devices. We have earned a reputation for manufacturing aneurysm prevention devices that perform at and above expectations, and meet CE Mark and FDA standards.
Etched Parts
Tecomet Inc.’s innovative processes provide us with the capabilities to produce parts with excellent feature and tolerance control, even with difficult-to-etch metals. We have developed a propriety process for chemically etching nitinol and titanium that cuts the metal with the tight tolerance and edge control required for medical devices. Parts manufactured by Tecomet Inc. are produced burr-free and have no heat-affected zones. We can chemically machine parts as thin as 0.0001 inches thick, up to 0.100 inches thick. Our production facilities include clean rooms and automated visual inspection equipment to ensure that the parts meet all industry, CE Mark and FDA standards. All our parts are manufactured to ISO 9001:2015.
At Tecomet Inc., we are committed to manufacturing the highest quality products, meeting or exceeding your expectations and delivering parts that will perform well in what can be life-saving applications. Contact us today!